MONOLITHOS, a company actively engaged in the ambitious ECO2Fuel project, has marked significant milestones in the field of electrocatalyst development and environmental sustainability. This article highlights the key achievements and challenges faced by MONOLITHOS in this groundbreaking endeavor.
Achievements of MONOLITHOS in ECO2Fuel
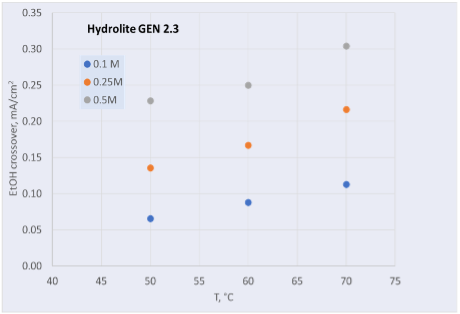
1. Stable Scaling of NiFeOx Electrocatalyst: A notable accomplishment is the successful scaling up of the NiFeOx electrocatalyst process. This scaling has remarkably maintained the structure, composition, morphology and performance of the electrocatalyst, ensuring its effectiveness and reliability.
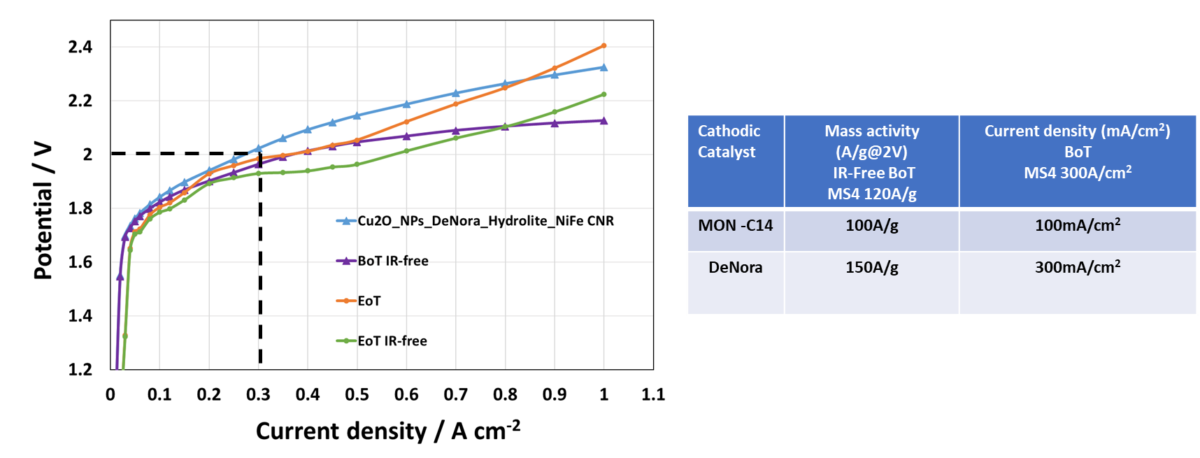
2. Development of Cu2O Electrocatalyst: The Cu2O electrocatalyst, developed under the ECO2Fuel project, has shown exceptional results. Its potential has been recognized with the decision to upscale its production, indicating its pivotal role in future applications.
3. Enhanced Anode Catalyst Activity: The company has fully achieved its milestone regarding the enhancement of anode catalyst activity. This improvement signifies a leap forward in the efficiency and effectiveness of the catalysts used.
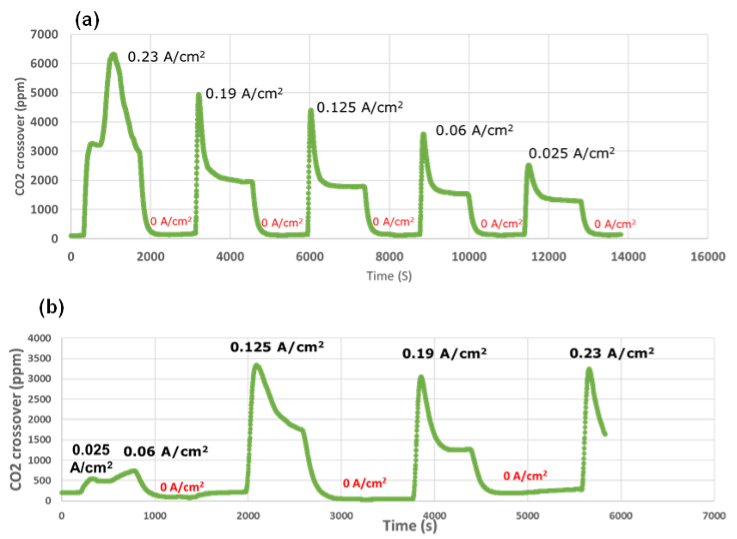
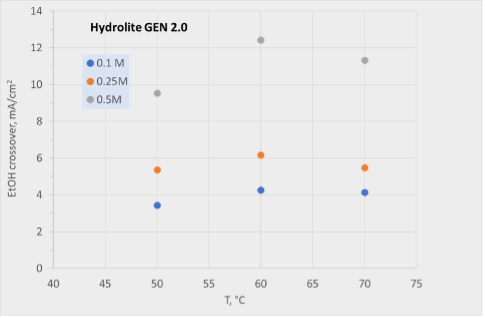
4. Progress in Cathode Catalyst Activity: The cathodic catalyst milestone has been successfully achieved by MONOLITHOS, demonstrating significant advancements in enhanced cathode catalyst activity. This progress is a testament to the company’s commitment to continuous improvement in catalyst development.
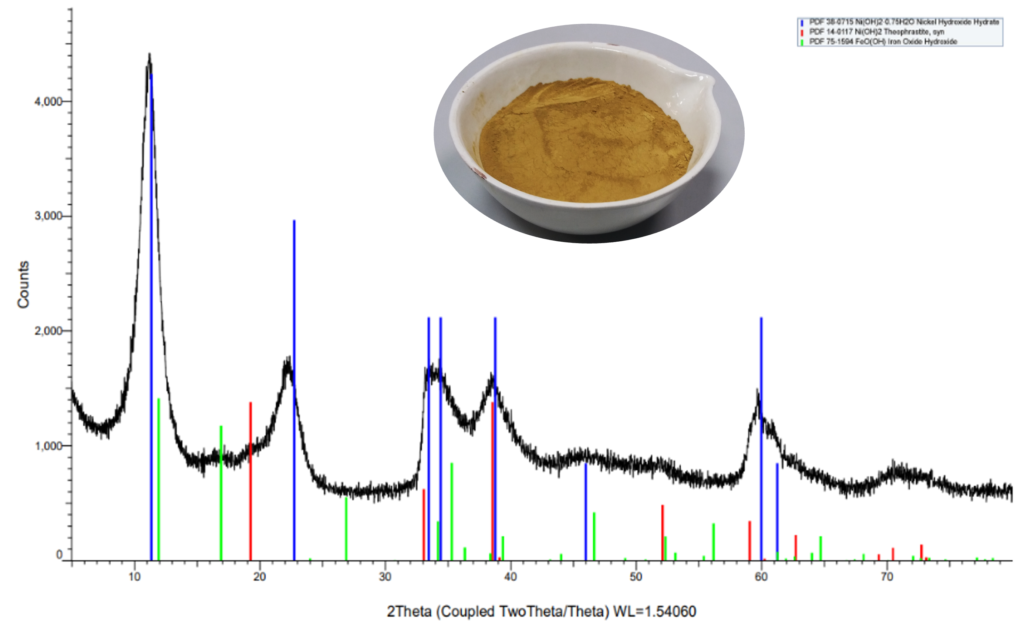
5. High Leaching Efficiencies: In an environmental triumph, the company has achieved over 99% leaching efficiencies for Cu and Ni from End-of-Life Membrane Electrode Assemblies (EoL MEAs). This was accomplished using an environmentally friendly hydrometallurgical leaching process, marking a significant step in sustainable practices.
6. Innovation with PtPd/CeZrO4 Catalyst: The synthesis of a PtPd/CeZrO4 catalyst through a wet impregnation process represents another innovative stride. This catalyst is set to be tested under simulated conditions involving diesel, biodiesel, and alcohol blends.



Challenges Faced by MONOLITHOS
Despite these achievements, MONOLITHOS faces certain challenges in the ECO2Fuel project:
1. Cathodic Performance Targets: One of the main challenges is achieving the high-performance targets set for the cathodic aspect of the project. Meeting these targets is crucial for the overall success and efficiency of the project.
2. Supply Chain Delays: There is a potential risk of delays in acquiring necessary materials, such as electrocatalyst precursors, equipment, and other essential components. These delays could impact the project timeline and its milestones.
Conclusion
MONOLITHOS’s involvement in the ECO2Fuel project has been marked by significant achievements, particularly in the development and scaling of innovative electrocatalysts and in advancing environmentally friendly processes. However, challenges such as meeting high-performance targets and potential supply chain delays pose hurdles that need to be navigated. The company’s continued dedication and innovative approach will be key in overcoming these challenges and achieving further success in sustainable energy solutions.
Source: MONOLITHOS